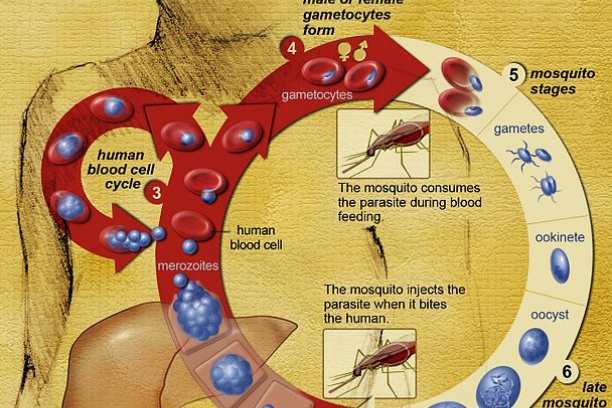
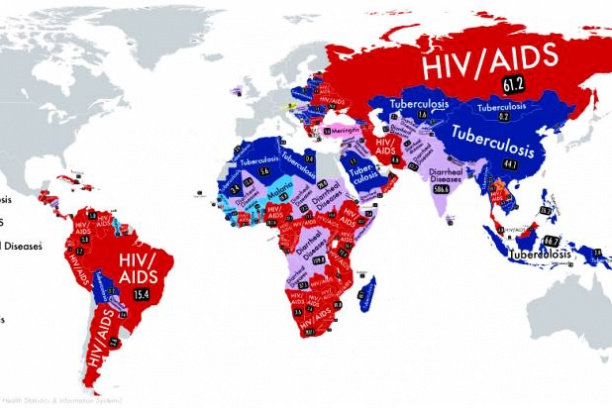
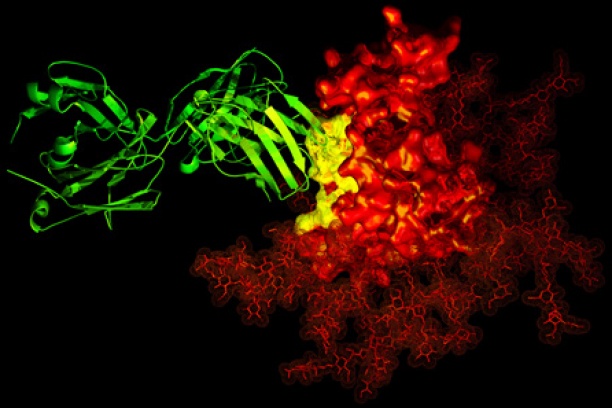
The goal of the Chemical Biology of Infectious Diseases (CBID) Training Program is to attract talented young people to the field and develop a new cadre of researchers prepared to address the growing threat of infectious diseases. Our Training Plan incorporates educating graduate students in key foundational concepts and tools and is fundamentally grounded in research. A significant new highlight of this program is that each CBID trainee will have a summer internship experience at a major pharmaceutical company that is actively engaged in antimicrobial drug discovery.